Toxicology in preclinical drug development 2013 is a critical phase that ensures the safety of novel therapeutic agents before human trials. This guide, presented by CONDUCT.EDU.VN, provides a detailed overview of toxicology studies, regulatory requirements, and best practices to support the development of safe and effective vaccines. Explore comprehensive toxicology assessments, risk mitigation strategies, and regulatory compliance, enhancing drug safety and efficacy.
1. Introduction to Preclinical Toxicology in Vaccine Development
Preclinical toxicology studies are essential for assessing the safety of new vaccines before they are tested in humans. These studies evaluate potential adverse effects of vaccines and adjuvants, ensuring that the benefits outweigh the risks. According to the World Health Organization (WHO), preclinical evaluations are crucial for identifying potential hazards and informing clinical trial design [1].
Vaccines, whether derived from recombinant forms, inactivated, or attenuated organisms, must meet stringent safety, quality, and efficacy requirements. Adjuvants, which enhance the immune response, are also subject to rigorous testing. Preclinical toxicology studies aim to identify any adverse effects and determine the safety profile of the vaccine candidate.
 Vaccine Development Timeline
Vaccine Development Timeline
2. Types of Preclinical Toxicology Studies
Several types of preclinical toxicology studies are conducted to evaluate vaccine safety, including single and repeat-dose toxicity, reproductive and developmental toxicity, mutagenicity, carcinogenicity, and safety pharmacology studies. These studies are essential to meet regulatory guidelines and ensure product safety. The Food and Drug Administration (FDA) requires comprehensive testing of vaccine components to identify any adverse effects [2].
2.1 Single and Repeat-Dose Toxicity Studies
Single and repeat-dose toxicity studies assess the acute and sub-acute effects of vaccine candidates. These studies help determine the dose range for subsequent studies and identify potential target organs for toxicity. They involve administering single or multiple doses of the vaccine to animals and monitoring for any adverse effects.
- Single-Dose Toxicity: Evaluates the effects of a single administration of the vaccine.
- Repeat-Dose Toxicity: Assesses the effects of multiple administrations over a specified period.
2.2 Reproductive and Developmental Toxicity Studies
Reproductive and developmental toxicity studies evaluate the potential effects of vaccines on fertility, pregnancy, and fetal development. These studies are particularly important for vaccines intended for use in women of childbearing potential. They assess potential effects on fertility, embryo-fetal development, and postnatal development.
- Fertility Studies: Assess the impact on reproductive function.
- Embryo-Fetal Development Studies: Evaluate the effects on the developing embryo and fetus.
- Postnatal Development Studies: Examine the effects on the offspring after birth.
2.3 Mutagenicity Studies
Mutagenicity studies assess the potential of vaccines to cause genetic mutations. These studies are generally not required for vaccines of biological origin but may be necessary for synthetic adjuvants. They typically involve in vitro assays to evaluate the potential for gene mutation, chromosome aberrations, and DNA damage.
2.4 Carcinogenicity Studies
Carcinogenicity studies evaluate the potential of vaccines to cause cancer. These studies are generally not required for vaccines due to the low dose and limited exposure, but may be considered for certain synthetic adjuvants.
2.5 Safety Pharmacology Studies
Safety pharmacology studies assess the potential effects of vaccines on physiological functions, such as the cardiovascular, respiratory, and central nervous systems. These studies are often integrated into repeat-dose toxicity studies.
- Cardiovascular System: Evaluates heart rate, blood pressure, and electrocardiogram.
- Respiratory System: Assesses respiratory rate, tidal volume, and hemoglobin oxygen saturation.
- Central Nervous System: Examines motor activity, behavior, coordination, and sensory/motor reflexes.
3. Factors Influencing Preclinical Toxicology Studies
Successful preclinical toxicology studies depend on several factors, including appropriate study designs, relevant animal models, and evoking an effective immune response. Additional in vivo and in vitro assays that establish the identity, purity, safety, and potency of the vaccine play a significant role in assessing critical characteristics of vaccine safety.
3.1 Study Design
The study design should be carefully planned to include the relevant species, route of administration, dose level, and number of doses. Control groups, including vehicle and adjuvant controls, are essential for accurate interpretation of the results. According to GLP guidelines, the study protocol should be written in accordance with regulatory requirements [3].
3.2 Animal Model Selection
Selecting the appropriate animal model is crucial for predicting human responses to vaccines. The animal model should mimic the human immune response and disease pathogenesis as closely as possible. Different animal models, such as mice, rabbits, and non-human primates, may be used depending on the specific vaccine and target disease.
3.3 Evoking an Effective Immune Response
The vaccine should evoke an effective immune response in the animal model to accurately assess its safety and efficacy. Immunogenicity testing, including antibody analysis and cell-mediated immunity assays, is essential for evaluating the immune response. The immune response should be robust enough to allow the detection of any adverse effects related to the vaccine or adjuvant.
4. Regulatory Requirements for Preclinical Toxicology Studies
Preclinical toxicology studies for vaccines must comply with regulatory requirements set forth by agencies such as the FDA and the European Medicines Agency (EMA). These regulations ensure that vaccines are safe and effective before they are tested in humans. Compliance with Good Laboratory Practice (GLP) guidelines is essential.
4.1 FDA Regulations
The FDA requires comprehensive preclinical testing of vaccine components to identify any adverse effects. These studies must follow GLP guidelines as described in the Code of Federal Regulations (CFR) 21. The toxicology studies must include single and repeat-dose toxicity, reproductive and developmental toxicity, mutagenicity, carcinogenicity, and safety pharmacology studies.
4.2 EMA Regulations
The EMA also requires preclinical testing of vaccines to ensure their safety and efficacy. The guidelines emphasize the importance of using appropriate animal models and study designs to predict human responses. The EMA provides detailed guidance on the types of studies required and the data that must be submitted for regulatory approval.
5. Key Endpoints in Preclinical Toxicology Studies
Preclinical toxicology studies evaluate various endpoints to assess the safety of vaccine candidates. These endpoints include clinical observations, body weight, food consumption, body temperature, injection site evaluation, ophthalmologic examination, clinical chemistry, hematology, urinalysis, bone marrow smears, C-reactive protein (CRP), creatine kinase, antibody analysis, necropsy, and histopathological evaluation.
5.1 Clinical Observations
Daily monitoring of animals for any signs of toxicity, including mortality, morbidity, general health, and behavior changes.
5.2 Body Weight and Food Consumption
Changes in body weight and food consumption can indicate adverse effects of the vaccine. These parameters reflect the overall health and well-being of the animals.
5.3 Body Temperature
Monitoring body temperature can help detect systemic inflammatory responses to the vaccine.
5.4 Injection Site Evaluation
Evaluation of the injection site for signs of inflammation, edema, erythema, and eschar formation. Draize scoring can be used to quantify these reactions.
5.5 Ophthalmologic Examination
Examination of the eyes to detect any signs of inflammation or toxicity, such as uveitis.
5.6 Clinical Chemistry
Analysis of blood samples to assess liver function, kidney function, electrolyte balance, carbohydrate metabolism, muscle enzymes, and protein levels.
5.7 Hematology
Analysis of blood samples to assess red blood cell parameters, white blood cell counts, and clotting parameters.
5.8 Urinalysis
Examination of urine to detect any abnormalities in physical appearance, specific gravity, microscopic appearance, and chemical composition.
5.9 Bone Marrow Smears
Analysis of bone marrow samples to evaluate hematopoiesis and detect any abnormalities in blood cell production.
5.10 C-Reactive Protein (CRP)
Measurement of CRP levels in blood as an indicator of systemic inflammatory response.
5.11 Creatine Kinase
Measurement of creatine kinase levels in blood as an indicator of muscle damage or inflammation.
5.12 Antibody Analysis
Measurement of antibody responses to assess the immunogenicity of the vaccine. ELISA and other methods are used to measure specific antibody levels.
5.13 Necropsy
Gross examination of all major organs to detect any abnormalities.
5.14 Histopathological Evaluation
Microscopic examination of tissue samples to detect any pathological changes. The site of vaccine injection and immune organs such as the spleen, thymus, and lymph nodes are carefully evaluated.
6. Special Considerations for Reproductive and Developmental Toxicology Studies
Reproductive and developmental toxicology studies are particularly important for vaccines intended for use in women of childbearing potential. These studies evaluate the potential effects of vaccines on fertility, pregnancy, and fetal development.
6.1 Study Design
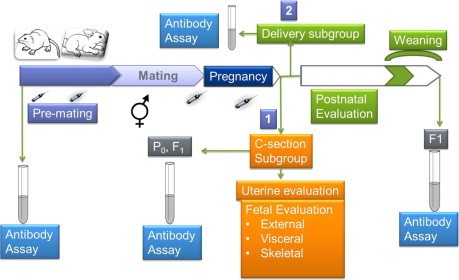
Reproductive studies should be designed following ICH S5(R2) guidelines. One species is required for this type of study. Animals should be immunized a few weeks before mating and boosted immediately prior to mating. Pregnant females are submitted to cesarean section and fetal examination on gestation day (GD) 18 for mice and on GD 20 for rats. Another subset is allowed to litter, and the post-natal development (PND) of the pups is followed up to weaning (rodent – PND 21). To assess the potential for long-lasting, permanent changes, the study could be extended to include assessment of the immune system (developmental immunotoxicity testing) in the offspring at 6–8 weeks.
6.2 FDA Pregnancy Categories
The FDA has assigned pregnancy categories to classify drugs and vaccines based on their potential risk to the fetus. These categories range from A (no risk demonstrated in well-controlled studies) to X (risk clearly outweighs any potential benefit).
- Category A: Adequate and well-controlled studies have failed to demonstrate a risk to the fetus.
- Category B: Animal reproduction studies have failed to demonstrate a risk to the fetus, and there are no adequate and well-controlled studies in pregnant women.
- Category C: Animal reproduction studies have shown an adverse effect on the fetus, but potential benefits may warrant use of the drug in pregnant women despite potential risks.
- Category D: There is positive evidence of human fetal risk, but potential benefits may warrant use of the drug in pregnant women despite potential risks.
- Category X: Studies in animals or humans have demonstrated fetal abnormalities, and the risks involved in use of the drug in pregnant women clearly outweigh potential benefits.
7. Specialized Toxicity Studies
For certain types of vaccines, specialized toxicity studies are needed, such as virulence and neurovirulence studies for live attenuated virus vaccines with a potential for reversion of attenuation or neurotropic activity. DNA vaccines also require bio-distribution studies to determine tissue distribution and potential for vector integration into the host genome.
7.1 Virulence and Neurovirulence Studies
These studies are required for new, live attenuated virus vaccines that have either a theoretical or an established potential for reversion of attenuation or neurotropic activity. Polio and yellow fever vaccines fall into this category. A neurovirulence test (NVT) for a polio vaccine is part of routine batch release procedures, and for yellow fever vaccine is designed to allow quantitative assessment of the effects of the virus by examination of specific areas following directed inoculation.
7.2 Bio-distribution Studies
For nucleic acid and viral vector-based vaccines, bio-distribution studies are necessary to determine the tissue distribution following administration and the potential for the vector to integrate into the host genome. These studies help assess the risk of off-target effects and potential long-term consequences.
8. Animal Models in Vaccine Research
Animal models play a critical role in vaccine research, providing valuable insights into disease pathogenesis, immune responses, and vaccine safety and efficacy. The selection of an appropriate animal model is essential for accurately predicting human responses to vaccines.
8.1 Considerations for Animal Model Selection
Criteria to consider when choosing animal models for vaccine development include similarities in immune organ development, transport of antibodies across mucosal surfaces, route of transmission, duration of immune memory, pattern of pathogenesis, receptors, the immune response ontogeny, lifespan and duration of neonatal, adolescent, and adult period, physiology (i.e., skin) for specific delivery, access to mucosal and systemic immune compartments, and transfer of passive immunity via the placenta, colostrum, and milk.
8.2 Examples of Animal Models
- Mice: Used to study acute extracellular bacterial infections and immune mechanisms.
- Guinea Pigs: Used to study tuberculosis and other infectious diseases due to their susceptibility and disease progression similar to humans.
- Rabbits: Used to study immunity to toxinogenic bacterial infections.
- Ferrets: Used to study influenza virus infections due to their similar respiratory tract physiology to humans.
- Non-Human Primates: Used to study HIV and other complex infectious diseases, providing the closest approximation to human immune responses.
9. Routes of Vaccine Administration
Selection of an appropriate route for vaccine administration is a critical component of a successful immunization. Vaccines are normally administered by injection; either intravenous (IV), intramuscular (IM), or subcutaneous (SC) administration. There are advantages and disadvantages for these routes of administrations. Vaccines could also be administered orally or intra-nasally, and these routes also have advantages and disadvantages.
9.1 Intramuscular (IM) Vaccine Administration
The needle used to administer the vaccine to the muscle should be long enough to reach deep into the muscle. It should be inserted at a 90° angle to the skin with a quick thrust. It is not necessary to aspirate when using this route. A minimum of 1 inch separation is necessary when using multiple injections in the same extremity. The following vaccines should be administered by the intramuscular (IM) route: Diphtheria-tetanus (DT, Td) with pertussis (DTaP, Tdap); Hemophilus influenzae type b (Hib); hepatitis A (HepA); hepatitis B (HepB); human papillomavirus (HPV); inactivated influenza (TIV); quadrivalent meningococcal conjugate (MCV4); and pneumococcal conjugate (PCV). Inactivated polio (IPV) and pneumococcal polysaccharide (PPSV23) could be administered either by IM or SC routes.
9.2 Subcutaneous (SC) Vaccine Administration
Subcutaneous tissue should be pinched up to prevent injection into muscle. The needle should be inserted at a 45° angle to the skin. It is not necessary to aspirate when using this route. A minimum of 1 inch separation is necessary when using multiple injections in the same extremity. The following vaccines should be administered by the SC route: measles, mumps, and rubella (MMR), varicella (VAR), meningococcal polysaccharide (MPSV4), and zoster (shingles [ZOS]).
9.3 Intranasal Vaccines
Intranasal vaccines can induce protective immunity in the respiratory tract were the viruses attack. By either slowing the rate of uptake of the antigens (e.g., intranasal vaccines are taken into the body more slowly than injectable vaccines, thus reducing the risk of allergic reaction) or by administering the vaccine viruses to an area of the body that they do not typically grow in (thus reducing the disease-causing effects of some of the strains of live vaccine viruses), the side-effects of the vaccine will be reduced. Intranasal administration is easy and acceptable to both humans and animals.
9.4 Alternative Routes
Few other non-injectable routes exist beside intranasal application. Orally- and intraperitoneally-administered vaccines (given into the abdominal cavity) have been investigated or approved for human use. These routes are used to improve the response of the gastrointestinal immune system to diseases like parvovirus and coronavirus. Polio vaccine, rotavirus, adeno or typhoid are examples of orally administered vaccines. Dermal patches, sprays (vaccines applied to the skin surface), and transdermal vaccines (aerosolized vaccine particles that are forced at high pressure through the skin using special instruments, thus avoiding the need for needles) have been developed. DNA plasmid vaccines are typically administered by the IM or ID route and may be given by electroporation which propels DNA-coated gold particles into various tissues.
10. Product Characterization
In addition to toxicity studies, in vivo and in vitro assays play a significant role in assessing critical safety characteristics of vaccines. Testing encompasses assessments for identity, purity, safety, and efficacy in terms of antigenicity and potency.
10.1 Key Assays
Among the most important are tests for potency, general safety (21CFR610.11), neurovirulence (IABS Scientific Workshop on Neurovirulence Tests for Live Vaccines, WHO, 2005), tumorigenicity (Meeting Report, WHO Study Group on Cell Substrates for Production of Biologicals, WHO, 2007; European Pharmacopoeia section 5.2.3), and pyrogenicity (21CFR610.13).
11. Pediatric Drug Development (Preclinical Safety Evaluations)
Pediatric evaluations are required as part of new drug and biologics licensing applications in the US and every marketing authorization application in Europe, unless a waiver has been granted. For juvenile toxicity studies (if pharmacological activity has been demonstrated), one species is acceptable. The rat is the recommended species (if relevant), because it has developmental systems which can be easily monitored. Other animal models could be used after careful consideration of its organ system development relative to that of humans.
12. Conclusion
Preclinical toxicology studies are a critical component of vaccine development, ensuring the safety and efficacy of novel therapeutic agents before human trials. By following regulatory requirements, selecting appropriate animal models, and conducting comprehensive toxicity assessments, researchers can minimize risks and develop safe and effective vaccines. For more detailed information and guidance on preclinical toxicology studies, visit CONDUCT.EDU.VN.
Ensure you are fully compliant and well-informed by consulting the expertise available at conduct.edu.vn. Contact us at 100 Ethics Plaza, Guideline City, CA 90210, United States, or via Whatsapp at +1 (707) 555-1234. Your path to safer vaccines starts here.
13. FAQ
-
What is the purpose of preclinical toxicology studies in vaccine development?
To assess the safety of new vaccines before they are tested in humans by evaluating potential adverse effects.
-
What types of preclinical toxicology studies are typically conducted?
Single and repeat-dose toxicity, reproductive and developmental toxicity, mutagenicity, carcinogenicity, and safety pharmacology studies.
-
Why is animal model selection important in preclinical toxicology studies?
The animal model should mimic the human immune response and disease pathogenesis as closely as possible to accurately predict human responses to vaccines.
-
What are the regulatory requirements for preclinical toxicology studies?
Compliance with regulatory requirements set forth by agencies such as the FDA and EMA, including Good Laboratory Practice (GLP) guidelines.
-
What are the key endpoints evaluated in preclinical toxicology studies?
Clinical observations, body weight, food consumption, body temperature, injection site evaluation, ophthalmologic examination, clinical chemistry, hematology, urinalysis, bone marrow smears, C-reactive protein (CRP), creatine kinase, antibody analysis, necropsy, and histopathological evaluation.
-
Why are reproductive and developmental toxicology studies important?
To evaluate the potential effects of vaccines on fertility, pregnancy, and fetal development, especially for vaccines intended for use in women of childbearing potential.
-
What are virulence and neurovirulence studies and when are they required?
These studies are required for new, live attenuated virus vaccines with a potential for reversion of attenuation or neurotropic activity to assess their safety.
-
What are bio-distribution studies and why are they necessary for DNA vaccines?
Bio-distribution studies determine the tissue distribution following administration and the potential for the vector to integrate into the host genome, assessing the risk of off-target effects and potential long-term consequences.
-
How does the route of vaccine administration affect the immune response?
The route of administration can influence the type and magnitude of the immune response, with intramuscular, subcutaneous, and intranasal routes each having their advantages and disadvantages.
-
What is the significance of product characterization in vaccine development?
Product characterization involves assessing the identity, purity, safety, and efficacy of vaccines, ensuring conformation to specified manufacturing standards and detecting undesirable contaminants or impurities.
References
- World Health Organization (WHO). (2005). WHO Guideline on Nonclinical Evaluation of Vaccines, Annex 1, WHO Technical Report Series No. 927.
- U.S. Food and Drug Administration (FDA). (2023). Code of Federal Regulations (CFR) 21.
- U.S. Food and Drug Administration (FDA). (2017). Good Laboratory Practice (GLP) Regulations.