Immunometabolism: A Guide to Interrogation. Immunometabolism, the convergence of immunology and metabolism, unveils the metabolic processes that intricately regulate immune cell function, impacting immune responses, inflammation, and disease progression. CONDUCT.EDU.VN offers comprehensive insights into immunometabolism and therapeutic targets. This guide will explore the methods and approaches to interrogate the field, ensuring a deeper understanding of its complexities, and explore the exciting world of immune cell metabolism and how it influences the fate of diseases like cancer.
1. Understanding the Foundations of Immunometabolism
Immunometabolism is the study of how metabolic processes regulate immune cell function. It explores the intricate relationship between metabolism and immunity, revealing how metabolic pathways influence immune cell development, activation, and function. This field is crucial for understanding how the body responds to pathogens, diseases, and therapies.
1.1. The Convergence of Immunology and Metabolism
Traditionally, immunology and metabolism were considered separate fields. However, it’s now clear that metabolic processes profoundly influence immune cell behavior. This convergence has led to the development of immunometabolism as a distinct area of study.
1.2. Key Concepts in Immunometabolism
Several key concepts underpin the field of immunometabolism:
- Metabolic Reprogramming: Immune cells undergo metabolic changes to meet the energetic and biosynthetic demands of their functions.
- Metabolic Checkpoints: These are regulatory points in metabolic pathways that influence immune cell activation and differentiation.
- Nutrient Sensing: Immune cells can sense changes in nutrient availability and adjust their metabolism and function accordingly.
1.3. Exploring Cellular Metabolism and Immunity
Cellular metabolism plays a crucial role in modulating the function of immune cells, influencing everything from their differentiation to their effector functions. In the rapidly evolving field of immunometabolism, researchers are uncovering new perspectives on how metabolism affects the immune system, which is essential for developing novel therapeutic strategies for various diseases.
2. Tools and Techniques for Studying Immunometabolism
Interrogating immunometabolism requires a variety of tools and techniques to analyze metabolic processes within immune cells. These methods range from basic biochemical assays to advanced imaging and computational approaches.
2.1. Metabolic Flux Analysis
Metabolic flux analysis (MFA) is a powerful technique for quantifying the rates of metabolic reactions within cells. It involves tracing the flow of labeled substrates through metabolic pathways to determine the activity of different enzymes and pathways.
- Isotope Tracing: Cells are incubated with labeled substrates, such as 13C-glucose or 15N-glutamine, and the incorporation of these isotopes into downstream metabolites is measured using mass spectrometry.
- Flux Calculation: Mathematical models are used to calculate the rates of metabolic reactions based on the isotope labeling patterns.
2.2. Seahorse XF Technology
Seahorse XF analyzers measure the oxygen consumption rate (OCR) and extracellular acidification rate (ECAR) of cells in real-time. These measurements provide insights into mitochondrial respiration and glycolysis, respectively.
- OCR Measurement: Indicates the rate of oxidative phosphorylation.
- ECAR Measurement: Reflects the rate of glycolysis.
- Metabolic Phenotyping: By manipulating culture conditions or using specific inhibitors, researchers can assess how different metabolic pathways contribute to immune cell function.
2.3. Mass Spectrometry-Based Metabolomics
Mass spectrometry-based metabolomics is used to identify and quantify a wide range of metabolites within cells. This approach provides a comprehensive snapshot of the metabolic state of immune cells under different conditions.
- Sample Preparation: Metabolites are extracted from cells using various methods, such as solvent extraction or protein precipitation.
- Mass Spectrometry Analysis: Metabolites are separated and detected based on their mass-to-charge ratio.
- Data Analysis: Statistical and bioinformatic tools are used to identify significant changes in metabolite levels.
2.4. Flow Cytometry and Imaging Techniques
Flow cytometry and imaging techniques are used to analyze metabolic parameters in individual immune cells. These methods allow researchers to study metabolic heterogeneity within cell populations and to correlate metabolic activity with other cellular characteristics.
- Flow Cytometry: Fluorescent probes are used to measure the levels of specific metabolites or metabolic enzymes in cells.
- Confocal Microscopy: High-resolution imaging can be used to visualize the localization of metabolites and metabolic enzymes within cells.
2.5. Gene Expression Analysis
Analyzing gene expression can provide valuable insights into the metabolic preferences and capacities of immune cells. This can be achieved through techniques like RNA sequencing (RNA-seq) and quantitative PCR (qPCR), which allow researchers to identify and quantify the expression levels of genes involved in various metabolic pathways.
2.6. Proteomic Analysis
Proteomic analysis complements gene expression studies by examining the actual protein levels and post-translational modifications of metabolic enzymes. Techniques such as mass spectrometry-based proteomics can identify changes in protein abundance, phosphorylation, and other modifications that affect enzyme activity and metabolic flux.
3. Immunometabolism in Different Immune Cell Types
Different immune cell types rely on distinct metabolic pathways to support their functions. Understanding these metabolic differences is crucial for developing targeted therapies that modulate immune responses.
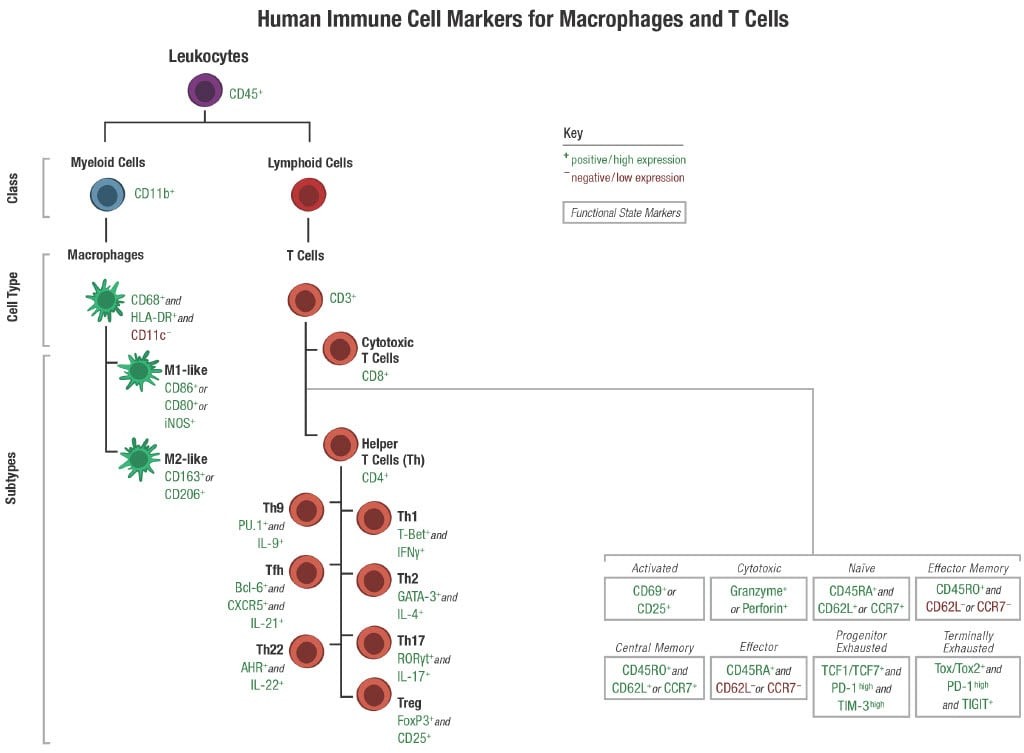
3.1. Macrophages
Macrophages are phagocytic cells that play a critical role in innate immunity. They can be broadly classified into two main subtypes: M1 (classically activated) and M2 (alternatively activated) macrophages.
- M1 Macrophages: Primarily rely on glycolysis for energy production and produce pro-inflammatory cytokines.
- M2 Macrophages: Utilize oxidative phosphorylation and fatty acid oxidation and promote tissue repair and immune regulation.
3.2. T Cells
T cells are lymphocytes that mediate adaptive immunity. They include several subtypes, such as CD4+ helper T cells and CD8+ cytotoxic T cells, each with unique metabolic requirements.
- CD4+ T Cells: Differentiate into various effector subsets, such as Th1, Th2, and Th17 cells, each with distinct metabolic profiles.
- CD8+ T Cells: Require glycolysis for rapid proliferation and cytotoxic activity.
- Regulatory T Cells (Tregs): Primarily rely on oxidative phosphorylation for their suppressive function.
3.3. Dendritic Cells
Dendritic cells (DCs) are antigen-presenting cells that initiate T cell responses. Their metabolic activity is critical for their ability to process and present antigens and to activate T cells.
- Glycolysis: Important for DC maturation and activation.
- Fatty Acid Oxidation: Required for DC migration and antigen presentation.
3.4. Neutrophils
Neutrophils are the most abundant type of white blood cells and are critical for the acute inflammatory response. They exhibit high glycolytic activity, which supports their rapid activation and migration to sites of infection.
- Glycolysis: Essential for neutrophil chemotaxis, phagocytosis, and production of reactive oxygen species (ROS).
- Pentose Phosphate Pathway (PPP): Provides NADPH, which is required for ROS production.
4. Immunometabolism in the Tumor Microenvironment
The tumor microenvironment (TME) is a complex ecosystem composed of cancer cells, immune cells, stromal cells, and extracellular matrix. Metabolic interactions within the TME significantly impact tumor progression and immune responses.
4.1. Metabolic Competition Between Cancer Cells and Immune Cells
Cancer cells often exhibit altered metabolism, such as increased glycolysis (the Warburg effect), which can deplete nutrients and create a hostile environment for immune cells.
- Glucose Deprivation: Cancer cells compete with immune cells for glucose, limiting their ability to function effectively.
- Lactate Accumulation: High glycolytic activity leads to the accumulation of lactate, which can suppress immune cell activity.
4.2. Metabolic Modulation of Immune Cell Function in the TME
The metabolic environment of the TME can directly influence the function of immune cells, promoting immune suppression and tumor evasion.
- Treg Recruitment and Activation: The TME can promote the recruitment and activation of Tregs, which suppress anti-tumor immune responses.
- Macrophage Polarization: The TME can skew macrophage polarization towards the M2 phenotype, which promotes tumor growth and angiogenesis.
4.3. Metabolic Checkpoints in Cancer Immunity
The metabolic checkpoint between glycolysis and the TCA cycle plays a pivotal role in regulating cellular function, particularly in adaptive immune responses and cancer progression. For example, phosphorylation of pyruvate dehydrogenase (PDH) by pyruvate dehydrogenase kinase (PDHK) is required for M1-like macrophage polarization. In the polarization process, expressions of PDHK1, PDHK3, and PDHK4 are elevated, leading to the phosphorylation of PDH and inhibition of its activity.
4.4. Therapeutic Strategies Targeting Immunometabolism in Cancer
Targeting metabolic pathways in cancer cells and immune cells can enhance anti-tumor immunity and improve therapeutic outcomes.
- Glycolysis Inhibitors: Drugs that inhibit glycolysis, such as 2-deoxyglucose (2-DG), can reduce the metabolic activity of cancer cells and enhance immune cell function.
- Fatty Acid Oxidation Inhibitors: Inhibiting fatty acid oxidation can disrupt the metabolic support of Tregs and M2 macrophages, promoting anti-tumor immunity.
- Metabolic Enhancers: Supplementing immune cells with specific metabolites, such as glutamine or arginine, can enhance their function and overcome metabolic suppression in the TME.
5. The Role of Glucose Metabolism in Cancer
Cancer cells exhibit distinct metabolic characteristics, with a prominent reliance on glycolysis even in the presence of oxygen, a phenomenon known as the Warburg effect. This metabolic shift supports rapid proliferation and tumor growth.
5.1. Glycolysis and the Warburg Effect
The Warburg effect is characterized by increased glucose uptake and glycolysis, even when oxygen is readily available. This metabolic reprogramming allows cancer cells to generate ATP and biosynthetic intermediates for rapid proliferation.
- Increased Glucose Transporters: Cancer cells upregulate the expression of glucose transporters, such as GLUT1, to enhance glucose uptake.
- Elevated Glycolytic Enzymes: Cancer cells increase the activity of glycolytic enzymes, such as hexokinase and pyruvate kinase, to accelerate glycolysis.
5.2. Regulation of Pyruvate Dehydrogenase (PDH)
Pyruvate dehydrogenase (PDH) is a key enzyme that links glycolysis to the tricarboxylic acid (TCA) cycle. Its activity is regulated by phosphorylation, with phosphorylation inhibiting PDH activity and promoting glycolysis.
- Pyruvate Dehydrogenase Kinase (PDK): PDK phosphorylates and inhibits PDH, diverting pyruvate away from the TCA cycle and towards lactate production.
- PDH Phosphatase (PDP): PDP dephosphorylates and activates PDH, promoting the TCA cycle and oxidative phosphorylation.
5.3. Lactate Production and the Tumor Microenvironment
Lactate is a byproduct of glycolysis that accumulates in the TME. It can suppress immune cell activity and promote tumor growth.
- Immune Suppression: Lactate inhibits the function of T cells and NK cells, reducing their ability to kill cancer cells.
- Angiogenesis: Lactate promotes angiogenesis, providing nutrients and oxygen to the growing tumor.
6. How the Metabolic Checkpoint Influences Immune Cell Types
The metabolic checkpoint between glycolysis and the TCA cycle influences different immune cell types and their functions. Pro-inflammatory immune cells such as M1-like macrophages and effector Th17 cells take up more glucose and enhance aerobic glycolysis. In contrast, anti-inflammatory immune cells such as M2-like macrophages and regulatory T cells use more oxidative phosphorylation.
6.1. M1-like Macrophages and M2-like Macrophages
Phosphorylation of pyruvate dehydrogenase (PDH) by pyruvate dehydrogenase kinase (PDHK) is required for M1-like macrophage polarization. In the polarization process, expressions of PDHK1, PDHK3, and PDHK4 are elevated, leading to the phosphorylation of PDH and inhibition of its activity. This metabolic change pivots cells from oxidative phosphorylation in mitochondria to glycolysis in the cytosol.
6.2. CD4+ Effector T Cells (Teff) and Regulatory T Cells (Treg)
Studies of CD4+ T cell populations in murine models show that PDHK1 is expressed in higher levels in the effector Th17 cells than in regulatory T cells. Inhibition of PDHK1 activity or downregulation of PDHK1 expression decreases glycolysis and reduces the effector Th17 cell proliferation, differentiation, and survival.
7. Immunometabolism and Therapeutic Advances
Understanding the intricacies of immunometabolism opens new avenues for therapeutic interventions, particularly in cancer and autoimmune diseases. By targeting metabolic pathways, it’s possible to modulate immune cell function and enhance therapeutic outcomes.
7.1. Targeting Metabolic Pathways in Cancer Therapy
Several therapeutic strategies aim to disrupt the metabolic support of cancer cells and enhance anti-tumor immunity.
- Inhibiting Glycolysis: Drugs like 2-deoxyglucose (2-DG) and lonidamine inhibit glycolysis, reducing the energy supply to cancer cells and making them more susceptible to immune attack.
- Modulating the TCA Cycle: Therapies that disrupt the TCA cycle can impair mitochondrial function and reduce ATP production in cancer cells.
- Targeting Fatty Acid Metabolism: Inhibitors of fatty acid synthesis and oxidation can disrupt lipid metabolism in cancer cells and immune cells, affecting tumor growth and immune responses.
7.2. Enhancing Immune Cell Metabolism
Strategies to boost the metabolic activity of immune cells can improve their ability to fight cancer and infections.
- Metabolic Supplementation: Providing immune cells with specific nutrients, such as glutamine or arginine, can enhance their metabolic activity and improve their function.
- Targeting Metabolic Checkpoints: Modulating metabolic checkpoints can redirect metabolic flux and enhance immune cell activation and effector function.
- Adoptive Cell Therapy: Genetically engineered immune cells can be designed to have enhanced metabolic capacity, improving their persistence and anti-tumor activity.
7.3. Clinical Trials and Future Directions
Several clinical trials are underway to evaluate the efficacy of immunometabolic therapies in cancer and other diseases. These trials are exploring various approaches, including metabolic inhibitors, immune cell enhancers, and combination therapies.
- Ongoing Clinical Trials: Evaluate the safety and efficacy of metabolic inhibitors in combination with chemotherapy or immunotherapy.
- Future Directions: Focus on developing more specific and effective metabolic modulators, as well as personalized approaches that tailor metabolic therapies to individual patients.
8. Case Studies in Immunometabolism
Real-world examples can illustrate the importance of immunometabolism in understanding and treating diseases. These case studies highlight the intricate interplay between metabolism and immunity in different contexts.
8.1. Immunometabolism in Sepsis
Sepsis is a life-threatening condition caused by a dysregulated immune response to infection. Metabolic alterations play a critical role in the pathogenesis of sepsis, affecting immune cell function and organ damage.
- Metabolic Alterations: Include increased glycolysis, impaired mitochondrial function, and altered amino acid metabolism.
- Therapeutic Strategies: Aim to restore metabolic homeostasis and improve immune function, such as using metabolic support and immunomodulatory therapies.
8.2. Immunometabolism in Autoimmune Diseases
Autoimmune diseases, such as rheumatoid arthritis and multiple sclerosis, are characterized by chronic inflammation and immune dysregulation. Metabolic abnormalities contribute to the development and progression of these diseases.
- Metabolic Abnormalities: Include altered glucose and lipid metabolism, as well as imbalances in cytokine production.
- Therapeutic Interventions: Focus on modulating metabolic pathways to reduce inflammation and restore immune tolerance.
8.3. Immunometabolism in Infectious Diseases
Infectious diseases trigger complex immune responses that are tightly regulated by metabolic processes. Understanding the metabolic requirements of immune cells during infection is crucial for developing effective therapies.
- Metabolic Responses: Include increased glycolysis and oxidative phosphorylation, as well as changes in amino acid and lipid metabolism.
- Therapeutic Approaches: Aim to support immune cell function and clear the infection, such as using metabolic enhancers and immunomodulatory agents.
9. Current Research and Breakthroughs in Immunometabolism
The field of immunometabolism is rapidly evolving, with new discoveries and breakthroughs emerging regularly. These advancements are providing deeper insights into the complex interplay between metabolism and immunity.
9.1. Recent Advances in Understanding Metabolic Pathways
Recent research has shed light on the roles of various metabolic pathways in regulating immune cell function.
- Pentose Phosphate Pathway (PPP): Critical for providing NADPH, which is required for antioxidant defense and biosynthesis.
- One-Carbon Metabolism: Involved in nucleotide synthesis and epigenetic regulation, influencing immune cell differentiation and function.
- Amino Acid Metabolism: Essential for protein synthesis, energy production, and signaling in immune cells.
9.2. New Therapeutic Targets and Strategies
Emerging therapeutic targets and strategies are being developed to modulate metabolic pathways and enhance immune responses.
- Targeting IDO1: Indoleamine 2,3-dioxygenase 1 (IDO1) is an enzyme that depletes tryptophan, suppressing T cell function in the TME. IDO1 inhibitors are being evaluated in clinical trials.
- Modulating Sirtuins: Sirtuins are a family of NAD+-dependent deacetylases that regulate metabolic and stress responses. Sirtuin activators are being explored as potential immunomodulatory agents.
9.3. Future Directions in Immunometabolism Research
Future research in immunometabolism will focus on several key areas:
- Personalized Immunometabolism: Tailoring metabolic therapies to individual patients based on their metabolic profiles.
- Systems-Level Analysis: Integrating multi-omics data to gain a comprehensive understanding of metabolic and immune interactions.
- Translational Studies: Moving basic research findings into clinical applications to improve patient outcomes.
10. Practical Applications of Immunometabolism Knowledge
The insights gained from immunometabolism research have numerous practical applications in various fields, from drug development to personalized medicine.
10.1. Drug Discovery and Development
Immunometabolism knowledge can be used to identify novel drug targets and develop more effective therapies for cancer, autoimmune diseases, and infectious diseases.
- Target Identification: Identifying metabolic enzymes or pathways that are critical for disease progression.
- Drug Screening: Screening compounds that modulate metabolic activity and enhance immune responses.
- Preclinical Studies: Evaluating the efficacy and safety of metabolic modulators in animal models.
10.2. Biomarker Discovery and Diagnostics
Metabolic biomarkers can be used to diagnose diseases, monitor treatment responses, and predict patient outcomes.
- Metabolite Profiling: Identifying metabolic signatures that are associated with disease states.
- Diagnostic Assays: Developing assays to measure metabolic biomarkers in patient samples.
- Personalized Medicine: Using metabolic biomarkers to tailor treatments to individual patients.
10.3. Nutritional Interventions and Lifestyle Modifications
Nutritional interventions and lifestyle modifications can be used to modulate metabolic pathways and improve immune function.
- Dietary Strategies: Modifying dietary intake to support immune cell metabolism and reduce inflammation.
- Exercise Programs: Engaging in regular exercise to improve metabolic health and immune function.
- Supplementation: Using specific nutrients or supplements to enhance immune cell activity.
By understanding how metabolism and immunity interact, researchers and clinicians can develop more effective strategies to prevent and treat a wide range of diseases.
FAQ: Immunometabolism
Here are ten frequently asked questions about immunometabolism, providing insights into this exciting field:
- What is immunometabolism?
Immunometabolism is the study of how metabolic processes regulate immune cell function. It explores the intricate relationship between metabolism and immunity, revealing how metabolic pathways influence immune cell development, activation, and function. - Why is immunometabolism important?
Understanding immunometabolism is crucial for developing new therapeutic strategies for various diseases, including cancer, autoimmune disorders, and infectious diseases. By targeting metabolic pathways, it’s possible to modulate immune cell function and enhance therapeutic outcomes. - What are the key concepts in immunometabolism?
Key concepts include metabolic reprogramming, metabolic checkpoints, and nutrient sensing. Metabolic reprogramming refers to the metabolic changes immune cells undergo to meet their energetic and biosynthetic demands. Metabolic checkpoints are regulatory points in metabolic pathways that influence immune cell activation and differentiation. Nutrient sensing involves immune cells detecting changes in nutrient availability and adjusting their metabolism and function accordingly. - How do different immune cell types use different metabolic pathways?
Different immune cell types rely on distinct metabolic pathways to support their functions. For example, M1 macrophages primarily rely on glycolysis, while M2 macrophages utilize oxidative phosphorylation. CD8+ T cells require glycolysis for rapid proliferation, while regulatory T cells rely on oxidative phosphorylation for their suppressive function. - What is the Warburg effect, and how does it relate to cancer?
The Warburg effect is characterized by increased glucose uptake and glycolysis in cancer cells, even when oxygen is readily available. This metabolic reprogramming allows cancer cells to generate ATP and biosynthetic intermediates for rapid proliferation. - How does the tumor microenvironment affect immunometabolism?
The tumor microenvironment (TME) is a complex ecosystem that includes cancer cells, immune cells, and stromal cells. Metabolic interactions within the TME significantly impact tumor progression and immune responses. For example, cancer cells can compete with immune cells for glucose, limiting their ability to function effectively. - What therapeutic strategies target immunometabolism in cancer?
Therapeutic strategies include inhibiting glycolysis with drugs like 2-deoxyglucose (2-DG), modulating the TCA cycle to impair mitochondrial function, and targeting fatty acid metabolism to disrupt lipid metabolism in cancer cells and immune cells. - How can nutritional interventions and lifestyle modifications improve immune function?
Nutritional interventions and lifestyle modifications can modulate metabolic pathways and improve immune function. Dietary strategies can support immune cell metabolism and reduce inflammation. Regular exercise can improve metabolic health and immune function. Specific nutrients or supplements can enhance immune cell activity. - What are some recent breakthroughs in immunometabolism research?
Recent advances include understanding the roles of the pentose phosphate pathway (PPP) and one-carbon metabolism in regulating immune cell function. Emerging therapeutic targets, such as IDO1, are being evaluated in clinical trials. - What are the future directions in immunometabolism research?
Future research will focus on personalized immunometabolism, systems-level analysis, and translational studies. Personalized immunometabolism involves tailoring metabolic therapies to individual patients based on their metabolic profiles. Systems-level analysis integrates multi-omics data to gain a comprehensive understanding of metabolic and immune interactions. Translational studies move basic research findings into clinical applications to improve patient outcomes.
Immunometabolism is a rapidly evolving field with significant implications for understanding and treating a wide range of diseases. By exploring the metabolic processes that regulate immune cell function, researchers and clinicians are paving the way for new therapeutic strategies and improved patient outcomes.
For more detailed information and guidance on immunometabolism, visit CONDUCT.EDU.VN. Our resources provide comprehensive insights and practical guidance for those seeking to understand and apply the principles of immunometabolism.
Navigating the complexities of immunometabolism can be challenging, but CONDUCT.EDU.VN is here to assist. We offer detailed guides and resources to help you understand and apply these principles in your research and practice. Contact us at 100 Ethics Plaza, Guideline City, CA 90210, United States. Whatsapp: +1 (707) 555-1234. Explore our website conduct.edu.vn for further reading and guidance.